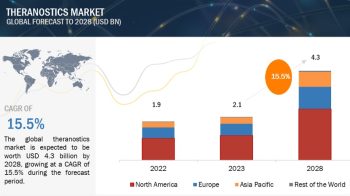
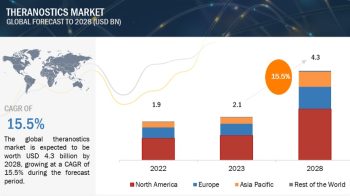
The report “Bioprocess Validation Market by Test Type (Extractables and Leachables, Integrity Testing, Microbiology Testing), Process Component (Filter Element, Bioreactors), End-User (CDMO, Biotechnology & Pharmaceutical Companies) – Global Forecast to 2024″, is exceeded USD 180 million in 2019 and is projected to be valued over USD 360 million by 2024, at a CAGR of 14.6% during the forecast period.
The stringent safety and quality regulations governing product certification and testing across the biopharmaceutical and pharmaceutical industries, high demand for the outsourcing of bioprocess validation services and rising R&D spending in the life science are some of the major factors that are expected to drive the growth of this market during the forecast period.
Additionally, automation in bioprocess validation services and geographic expansions by market players are the upcoming trends in bioprocess validation, which also expected to support market growth in the coming years.
Download PDF Brochure@
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=39981433
Recent Developments:
# In August 2018, Merck KGaA launched the BioReliance Product Characterization Portfolio to enhance its service portfolio in the bioprocess validation market.
# In March 2017, Toxikon and SG Lab (Italy) entered into a partnership, to extend its analytical and extractable & leachable testing service portfolio to determine the drug integrity in the entire life cycle.
# In January 2019, SGS acquired LeanSis Expertos en Productividad S.L. (Spain). This acquisition provided SGS operational and manufacturing training and capacity building services to over 200 clients across Spain.
# In February 2018, Pall Corporation expanded its Hoegaarden office in Belgium with new R&D center, customer demonstration labs, and manufacturing space. This will help the company to establish its position in the Belgium market.
Contract Development and Manufacturing Organizations (CDMOs) to form the fastest-growing end-user segment in the bioprocess validation market
Among all end-user segments, the demand for bioprocess validation is estimated to grow at the highest rate among CDMOs. This can majorly be attributed to the stringent FDA safety regulations and consumer lawsuits regarding testing requirement, accelerating costs of validation and quality check processes after or during drug development, increasing adoption of bioprocess validation services to keep biopharma production, and approvals in line with the regulatory requirements.
North America is estimated to be the largest regional market for bioprocess validation
North America is expected to account for the largest share of the bioprocess validation market in 2019, closely followed by Europe. Factors such as the presence of well-established biopharmaceutical industry in the region, high R&D expenditure, presence of leading bioprocess validation service providers, and stringent regulatory requirements for biopharmaceutical production are driving the growth of the bioprocess validation market in North America.
Critical questions the report answers:
# Where will all these developments take the industry in the medium-to-long-term?
# What are the major trends in bioprocess validation?
# Who are the major end-users of bioprocess validation?
# Which test types are used mainly for bioprocess validation?
# Which process component has a higher adoption rate of bioprocess validation?
Request Sample Pages@
https://www.marketsandmarkets.com/requestsampleNew.asp?id=39981433
The prominent players operating in the bioprocess validation market are Merck KGaA (Germany), Sartorius Stedim Biotech (Sartorius AG) (France), Pall Corporation (US), SGS S.A. (Switzerland), Eurofins Scientific (Luxembourg), Cobetter Filtration Equipment Co., Ltd. (China), Toxikon Corporation (US), DOC S.r.l. (Italy), MEISSNER FILTRATION PRODUCTS, INC. (US), and Thermo Fisher Scientific (US).