Overview of This Study:
This study involved the extensive use of primary and secondary sources and involved an in-depth analysis of the various factors affecting the growth of the global Bioprocess Validation Research Technique Market. Both primary and secondary research was used to identify the segmentation types, trends, key players, key market dynamics, and key player strategies as well as derive the competitive leadership mapping and competitive landscape in this market.
Expected Revenue Growth:
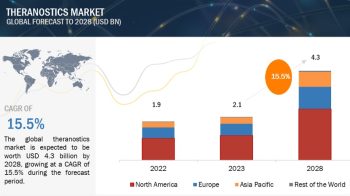
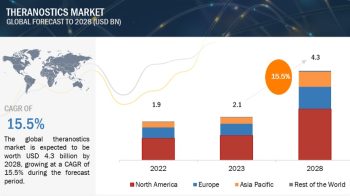
The global bioprocess validation market is exceeded USD 180 million in 2019 and is projected to be valued over USD 360 million by 2024, at a CAGR of 14.6% during the forecast period.
Global Growth Boosting Factors:
Stringent safety and quality regulations governing product certification and testing across the biopharmaceutical and pharmaceutical industries, high demand for the outsourcing of bioprocess validation services, and regulatory mandates in the healthcare industry to maintain compliance with Good Manufacturing Practices (GMP) are the major factors driving the growth of this market.
Download PDF Brochure@
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=39981433
Objectives of the Study:
# To define, describe, and forecast the Bioprocess Validation Research Technique Market based on test type, process component, end-user, and region.
# To provide detailed information regarding the factors influencing market growth (such as drivers, restraints, opportunities, challenges, and industry trends)
# To analyze micromarkets with respect to individual growth trends, prospects, and contributions to the overall bioprocess validation services market.
# To analyze market opportunities for stakeholders and provide details of the competitive landscape for key players operating in this market.
# To strategically profile the key players in the bioprocess validation services market and comprehensively analyze their market shares and core competencies
# To strategically analyze five different geographical regions, namely, North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
# To track and analyze competitive developments such as joint ventures & acquisitions; product developments and enhancements; agreements & partnerships; expansions; and funding activities in the bioprocess validation market.
End Users:
Contract Development and Manufacturing Organizations (CDMOs) to form the fastest-growing end-user segment in the bioprocess validation market
Among all end-user segments, the demand for bioprocess validation is estimated to grow at the highest rate among CDMOs. This can majorly be attributed to the stringent FDA safety regulations and consumer lawsuits regarding testing requirement, accelerating costs of validation and quality check processes after or during drug development, increasing adoption of bioprocess validation services to keep biopharma production, and approvals in line with the regulatory requirements.
Geographical Growth Scenario:
North America is estimated to be the largest regional market for bioprocess validation
North America is expected to account for the largest share of the Bioprocess Validation Research Technique Market in 2019, closely followed by Europe. Factors such as the presence of well-established biopharmaceutical industry in the region, high R&D expenditure, presence of leading bioprocess validation service providers, and stringent regulatory requirements for biopharmaceutical production are driving the growth of the bioprocess validation market in North America.
Request Sample Pages@
https://www.marketsandmarkets.com/requestsampleNew.asp?id=39981433
Global Leaders:
The prominent players operating in the bioprocess validation market are Merck KGaA (Germany), Sartorius Stedim Biotech (Sartorius AG) (France), Pall Corporation (US), SGS S.A. (Switzerland), Eurofins Scientific (Luxembourg), Cobetter Filtration Equipment Co., Ltd. (China), Toxikon Corporation (US), DOC S.r.l. (Italy), MEISSNER FILTRATION PRODUCTS, INC. (US), and Thermo Fisher Scientific (US).