Factors such as the growing worldwide prevalence of HIV/AIDS, increasing number of blood transfusions and blood donations, benefits offered by point-of-care instruments and kits, and increasing government initiatives are expected to drive the growth of the market worldwide.
The HIV diagnosis market is projected to reach USD 3.88 Billion by 2021 from USD 2.35 Billion in 2016, at a CAGR of 10.5% from 2016 to 2021. HIV Diagnosis Market by Test Type (Antibody (ELISA, Rapid, Western Blot), Viral Load, CD4 Count, Early Infant, Viral Identification, Product (Assay, Kit, Reagent, Instrument, Software, Services), End User (Hospitals) – Global Forecast to 2021.
AIDS is a major public health concern with a prevalence rate that has increased dramatically in the past two decades. This increase in the prevalence of HIV, and the subsequent increase in the number of patients suffering from AIDS, has resulted in an increase in the demand for advanced technologies for HIV diagnostics.
According to the World Health Organization (WHO), in 2016, around 36 million people were diagnosed with HIV globally, of which 2.1 million were reported to be cases of new HIV infections. Globally, 1.1 million people died from HIV-related causes in 2015. According to the CDC, in 2015, more than 1.2 million people in the U.S. were reportedly infected with HIV, and almost 19% of these were not aware about their status. According to Canada’s source for HIV, the CATIE, in 2014, 75,500 Canadians were found to be HIV-infected, and HIV incidence has increased by 9.7% since 2011. According to the Robert Koch Institute, there were 84,700 individuals with HIV in Germany in 2016 and more than 3,200 new cases of HIV in 2015. According to AVERT, in 2015, 5.1 million people were living with HIV/AIDS in Asia, of which only 64% population were aware about their HIV status. In the same year, 180,000 people died of AIDS in Asia.
This increasing prevalence of HIV across the globe will result in an increased need for diagnostic and testing procedures, which in turn will boost the HIV diagnostics market growth.
Download PDF Brochure:
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=66008285
Point-of-care (PoC) HIV tests are also known as rapid HIV tests. In regular practice, HIV diagnosis includes pre-test counselling, HIV testing, and post-test counselling wherein patients are required to return for test results at later dates. In PoC HIV diagnosis, counselling and screening are carried out simultaneously while the patient is still in the lab. This is followed by confirmatory tests, if needed. Since pre-test and post-test counselling are carried out at the same in PoC tests, individuals are not required to return for their results, making it a very convenient option.
Rapid HIV tests provide results in less than 30 minutes, eliminating the stress of waiting for results. These tests detect antibodies in the serum/blood but the detection window is 3–4 weeks. Rapid HIV testing mainly targets undiagnosed cases for HIV as well as those who have not returned for follow-up testing. As a result of their various advantages, the demand for technologically advanced PoC kits and instruments is on the rise, which is expected to aid in the growth of the HIV diagnostics market in the coming years.
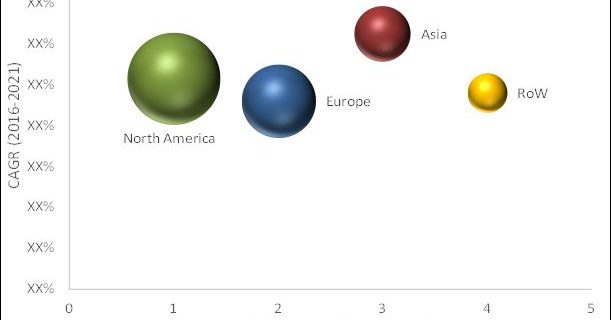
Region Covered in HIV Diagnostics Market
The global HIV diagnosis market is segmented into four major regions—North America, Europe, Asia, and Rest of the World. Geographically, the market is dominated by North America. Growth in the North American segment is primarily driven by the high incidence and prevalence of HIV/AIDS, increasing awareness about HIV/AIDS, and growing government initiatives and increasing blood donations.
Request for Sample Pages:
https://www.marketsandmarkets.com/requestsampleNew.asp?id=66008285
Leading Companies in HIV Diagnostics Market
Key players in the HIV Diagnosis Market are Siemens AG (Germany), Roche Diagnostics (Switzerland), Abbott Laboratories (US), Danaher Corporation (US), Thermo-Fisher Scientific Inc. (US), Merck KGaA (Germany), Becton, Dickinson and Company (US), Hologic, Inc. (US), Alere Inc. (US), and Bio-Rad Laboratories (US).