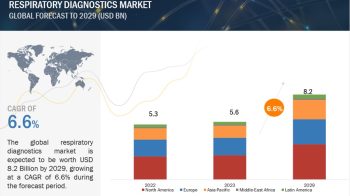
The in vitro diagnostics (IVD) quality controls market is broadly categorized into seven application segments—clinical chemistry, immunochemistry, molecular diagnostics, hematology, microbiology, coagulation/hemostasis, and other applications. Among these, the immunochemistry segment accounted for the largest share of the IVD quality control applications market in 2015. This is primarily because immunoassay tests hold a large share in the IVD market and the market is witnessing a growing adoption of multi-analyte controls in immunoassay tests. However, the molecular diagnostics segment is expected to witness the highest growth in the application market, majorly due to the rising prevalence of infectious diseases.
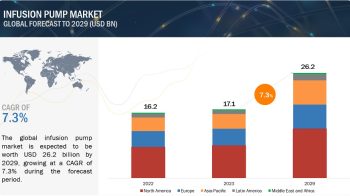
Based on manufacturer type, the IVD quality controls market is broadly segmented into instrument manufacturer controls and independent manufacturer controls; the independent manufacturer controls segment is subsegmented into third-party controls and instrument-specific controls. Third-party controls accounted for the largest market share in 2015 primarily due to the increasing use of these controls across the globe to verify patient test results. Moreover, this segment is expected to witness the highest growth in the coming years due to the rising use of third-party controls among laboratories as a result of recommendations from regulatory bodies to use these controls over the ones offered by instrument or reagent manufacturers.
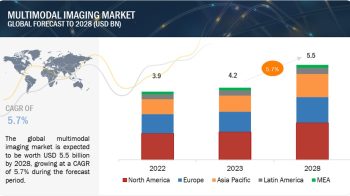
In 2015, North America commanded the largest share of the IVD quality control products market, followed by Europe. Although North America will continue to be the largest market for IVD quality control products, the Asia-Pacific region is expected to witness the highest growth during the forecast period. The high growth in the Asia-Pacific market can be attributed to the improving healthcare infrastructure, growing IVD test volumes, rapid growth in the number of accredited laboratories, and increasing use of quality control products by laboratories operating in this region. Moreover, in some countries such as Australia, New Zealand, and Taiwan, ISO 15189 (Medical laboratories – Requirements for quality and competence) accreditation has become mandatory, which is expected to drive the demand for IVD quality controls in these countries.