Overview of This Study:
This study involved four major activities in estimating the current size of the clinical trial supplies market. Exhaustive secondary research was carried out to collect information on the market, its peer markets, and its parent market. The next step was to validate these findings, assumptions, and sizing with industry experts across the value chain through primary research. Both top-down and bottom-up approaches were employed to estimate the complete market size. After that, market breakdown and data triangulation procedures were used to estimate the size of segments and subsegments.
Market Size Estimation:
Both top-down and bottom-up approaches were used to estimate and validate the total size of the market. These methods were also used extensively to estimate the size of various subsegments in the market. The research methodology used to estimate the market size includes the following:
# The key players in the industry have been identified through extensive secondary research
# The revenues generated by leading players operating in the clinical trial supplies market have been determined through primary and secondary research
# All percentage shares, splits, and breakdowns have been determined using secondary sources and verified through primary sources
Download PDF Brochure@
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=172240059
Logistics services segment to dominate the clinical trial supplies market in 2020
Based on service, the clinical trial supplies market is segmented into logistics; distribution, storage, and retention; packaging, labeling, and blinding; manufacturing; comparator sourcing; and solutions. The logistics services segment is expected to account for the largest share of this market in 2020, primarily due to the globalization of clinical trials and the increasing number of trials involving temperature-sensitive products.
Recent Developments in the Global Market:
# In December 2019, Catalent launched the New CTSuccess Service to guide sponsors toward smarter clinical trial supply planning.
# In May 2019, Catalent completed the USD 1.2-billion acquisition of Paragon Bioservices, Inc., a leading viral vector development and manufacturing partner for gene therapies.
# In September 2019, Catalent announced that it would invest USD 9 million in a new clinical supply facility in San Diego, due to open in the summer of 2020.
Phase III segment to register the highest growth in the clinical trial supplies market, by phase
Based on phase, the clinical trial supplies market is segmented into phase I, phase II, phase III, phase IV, and BA/BE studies. The phase III segment is expected to account for the largest share of the clinical trial supplies market in 2020. This segment is also estimated to grow at the highest CAGR during the forecast period. The large share and high growth of this segment can be attributed to the fact that phase III trials are the most expensive, time-consuming, and difficult to design and run.
Key Questions Addressed by the Report:
# Where will all these developments take the industry in the mid-to-long term?
# What are the recent trends affecting the market?
# Who are the key players in the market, and how intense is the competition?
# Which region is a lucrative market for clinical trial supplies?
Request Sample Pages@
https://www.marketsandmarkets.com/requestsampleNew.asp?id=172240059
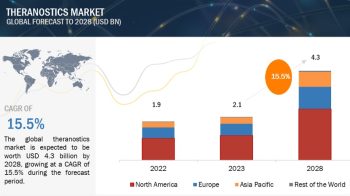
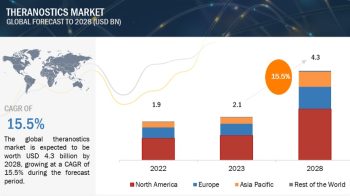
North America is the largest regional market for clinical trial supplies
The global clinical trial supplies market is segmented into North America, Europe, the Asia Pacific, and the Rest of the World. North America is the largest regional market for clinical trial supplies, followed by Europe. The large share of the North American market can primarily be attributed to the presence of well-established CROs and the high and growing R&D expenditure by pharmaceutical and biopharmaceutical companies in the region.
Global Key Players:
The prominent players operating in the global clinical trial supplies market include Alamc Group (UK), Catalent, Inc. (US), PCI Services (US), Parexel International Corporation (US), Sharp Packaging Services (US), Biocair (UK), O&M Movianto (US), KLIFO A/S (Denmark), and Thermo Fisher Scientific, Inc. (US).