What This Report Will Provide?
The growth of this market is majorly driven by the increased utilization of the low priced reprocessed medical devices as compared to new devices, increased efforts towards reducing regulated medical waste (RMW), and rising number of surgical procedures associated with the growing prevalence of chronic diseases. However, the risk of transmission of surgical site infections associated with the use of reprocessed devices may hinder the growth of the market to a certain extent.
Expected Revenue Growth:The
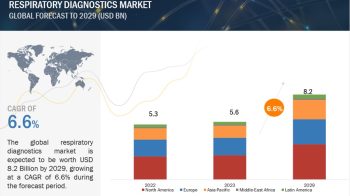
Global medical device reprocessing market is projected to be valued at USD 823.5 Million in 2017 and is expected to grow at a CAGR of 16.3% during the forecast period to reach to USD 1,754 Million by 2022. Base year considered for the report is 2016.
Major Growth Boosters:
The medical device reprocessing includes cleaning, disinfection, inspection, sterilization, and repackaging of used medical devices for its reuse to diagnose and treat multiple patients.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=583
Objectives of the Study
- To define, describe, and forecast the global market with respect to product & service, type of device, application, and region
- To provide detailed information regarding the major factors influencing the growth of the market (drivers, restraints, opportunities, and challenges)
- To strategically analyze micromarkets with respect to individual growth trends, prospects, and contributions to the overall market
- To analyze market opportunities for stakeholders and provide details of the competitive landscape of market leaders
- To forecast the size of the market with respect to four main regions—North America, Europe, Asia-Pacific, and the Rest of the World
Target Audience:
- Third-party medical device reprocessors and distributors
- Hospitals, clinics, and other healthcare laboratories
- Academic and research institutions
- Government associations
- Market research and consulting firms
- Venture capitalists and investors
Request Sample Report: https://www.marketsandmarkets.com/requestsampleNew.asp?id=583
The reprocessing practices are regulated by various regional regulatory authorities. The changing regulatory scenario favoring medical device reprocessing in a number of countries, such as France and Japan, will open up new areas of opportunity for market players. For example, till 2016, the outsourcing of medical device reprocessing was prohibited in France; however, the European Union (EU) Regulations (Article 17), released on April 5, 2017, mandated the use of third-party reprocessing services by all French hospitals.
Regional Growth Analysis:
Geographically, the global medical device reprocessing market is segmented into North America, Europe, Asia-Pacific, and the Rest of the World. In 2016, North America dominated the market, and this is primarily attributed to its well-established healthcare industry, growing prevalence of chronic diseases, and increasing volume of surgical procedures. The European region is expected to witness the highest CAGR during the forecast period, owing to the rising prevalence of chronic diseases, increasing geriatric population, rising healthcare expenditure, increasing number of surgical procedures, and growing awareness about the usage of reprocessed medical devices