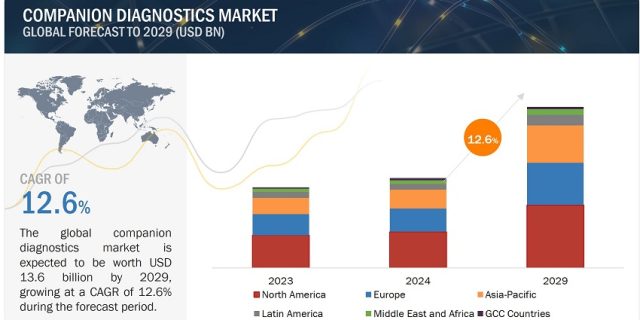
The companion diagnostics industry is experiencing remarkable growth, projected to soar from $7.5 billion in 2024 to $13.6 billion by 2029, at an impressive compound annual growth rate (CAGR) of 12.6%. This surge can be attributed to the increasing significance of personalized medicine, the rising prevalence of chronic diseases like cancer, and the growing adoption of targeted therapies.
Download a PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=155571681
Personalized Medicine: The Driving Force
The increasing awareness of the advantages of personalized medicine is a key driver propelling the utilization of companion diagnostics and genomics & proteomics technologies. These diagnostic tests analyze specific indications for particular drugs in individual patients, enabling healthcare providers to devise tailored treatment plans and minimize the risk of adverse events. The shift towards personalized medicine is reshaping healthcare practices, with a focus on delivering more precise and effective treatments, further fueling the demand for companion diagnostics products.
Opportunities and Challenges
While the increasing importance of companion diagnostics in drug development presents a significant opportunity for market growth, the industry faces challenges. The shortage of adequately trained professionals in companion diagnostic testing poses a hurdle, as these complex technologies require skilled personnel for accurate testing and interpretation.
Market Landscape
The companion diagnostics market is highly consolidated, with major players like F. Hoffmann-La Roche Ltd., Agilent Technologies, Inc., QIAGEN, Thermo Fisher Scientific, Inc., and Abbott Laboratories, Inc. dominating the scene. These industry giants leverage their extensive product portfolios, robust marketing and distribution networks, and substantial R&D budgets to maintain their market dominance, creating barriers for new entrants.
Product and Technology Landscape
In 2023, the assays, kits & reagents segment held the largest market share, driven by the wide variety of available products and their growing utilization across various therapeutic areas. The polymerase chain reaction (PCR) technology segment also captured a significant portion of the market due to its ease of use and the extensive availability of PCR kits & reagents for companion diagnostic testing.
Cancer: A Significant Market Driver
The cancer segment held the largest share of the companion diagnostics market in 2023, fueled by the increasing role of companion diagnostics in personalized cancer treatments and the growing significance of biomarkers in cancer diagnosis. Additionally, tissue samples dominated the sample type segment, as they enable direct analysis of tumors, providing a comprehensive view of genetic mutations and biomarkers for more accurate and personalized treatment decisions.
Regional Landscape
North America, led by the United States and Canada, emerged as the largest regional market for companion diagnostics in 2023. The region’s technological advancements and a high number of product approvals by regulatory authorities like the FDA have driven market growth.
As the companion diagnostics industry continues to evolve, it plays a pivotal role in shaping the future of personalized medicine, enabling healthcare providers to make informed treatment decisions and ultimately improving patient outcomes.