The Research Report on“Embolic Protection Devices Market by Type (Distal Filter, Distal Occlusion, Proximal Occlusion), Material (Nitinol, Polyurethane), Application (Cardiovascular, Neurovascular, Peripheral), Indication (PCI, SVD, TAVR) – Global Forecast to 2023″ published by MarketsandMarkets™, is projected to reach USD 604.9 Million, at a CAGR of 8.3% during the forecast period.
The growth in this market is mainly attributed to factors such as the rising incidence of cardiovascular and neurovascular diseases, growing funding and investments, launch of technologically advanced embolic protection devices, and the rising demand for minimally invasive procedures.
Research Methodology;
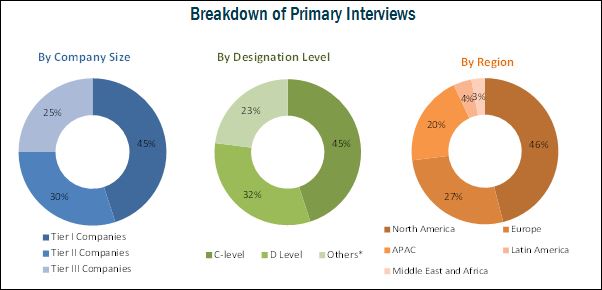
Top-down and bottom-up approaches were used to validate the size of the global market and estimate the size of various other dependent submarkets. Major players in the market were identified through secondary sources; directories; databases such as Hoovers, Bloomberg Businessweek, Factiva, and OneSource, journals and their market revenues were determined through primary and secondary research. Secondary research included the study of the annual and financial reports of top market players, whereas primary research included extensive interviews with the key opinion leaders such as CEOs, directors, and marketing executives. The percentage splits, shares, and breakdowns of the product markets were determined using secondary sources and verified through primary sources. All possible parameters that affect the market covered in this research study have been accounted for, viewed in extensive detail, verified through primary research, and analyzed to get the final quantitative and qualitative data.
Request Research Sample Pages:
https://www.marketsandmarkets.com/requestsampleNew.asp?id=158143803

Embolic Protection Devices Market
Embolic Protection Devices Market Segmentation in Detailed:
The neurovascular segment is expected to grow at the highest CAGR during the forecast period
On the basis of application, categorized into cardiovascular, neurovascular, and peripheral vascular diseases. The neurovascular segment is expected to grow at the highest CAGR during the forecast period. The favorable medical reimbursement scenarios for neurovascular procedures in developed countries, increasing awareness about the clinical efficacy of embolic protection devices among neurosurgeons, and the rising adoption of these systems across developing countries (such as China and India) are expected to drive the growth of this application segment during the forecast period.
The percutaneous coronary intervention segment accounted for the largest market share in 2017
On the basis of indication, categorized into percutaneous coronary intervention, transcatheter aortic valve replacement (TAVR), carotid artery diseases, saphenous vein graft disease, and other indications. In 2017, the percutaneous coronary intervention segment accounted for the largest share of the global embolic protection devices market. The large share of this segment can primarily be attributed to the increasing prevalence of CAD and the rising preference for minimally invasive procedures.
Geographical View in-detailed:
North America dominated the embolic protection devices market. The rising geriatric population and prevalence of cardiovascular and neurovascular diseases, increasing adoption of minimally invasive techniques, increasing availability of funding, and the presence of a large number of medical device product manufacturers are driving the growth of the market in North America. However, the APAC is expected to grow at the highest CAGR during the forecast period. The high growth of this segment is attributed to the growing geriatric population, rising prevalence of CVD, increasing number of facilities, and rising healthcare expenditure.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=158143803
Global Key Leaders:
The prominent players in the embolic protection devices market are Boston Scientific (US), Medtronic (Ireland), Abbott (US), Cordis (A Cardinal Health Company) (US), Allium Medical (Israel), Contego Medical (US), W.L. Gore & Associates (US), Silk Road Medical (US), Claret Medical (US), and AngioSlide (Israel).