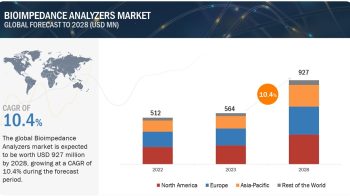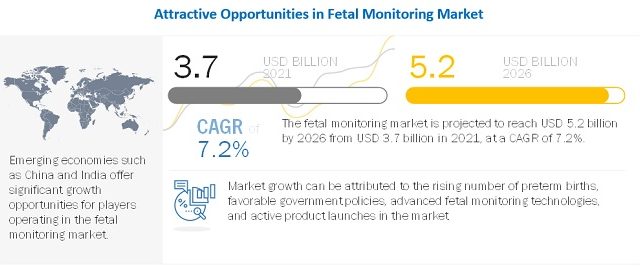
The fetal monitoring market is projected to reach USD 5.2 billion by 2026 from USD 3.7 billion in 2021, at a CAGR of 7.2% from 2021 to 2026.
The growth of this market is driven by the escalating prevalence of preterm births, favorable government policies, advanced technologies of fetal monitoring systems, and active product launches.
“Ultrasound Devices is expected to hold the largest share of the fetal monitoring market, by product in 2021.”
Based on products, the fetal monitoring market is segmented into ultrasound devices, electronic maternal/fetal monitors, fetal electrodes, fetal doppler devices, uterine contraction monitors, telemetry devices, accessories & consumables, and other products. Ultrasound devices are the largest segment in this market. This segments growth can primarily be attributed to technological advancements, increasing congenital anomalies, maternal mortality, and rising regulatory approvals.
To know about the assumptions considered for the study, Request for Free Sample Report @ https://www.marketsandmarkets.com/requestsampleNew.asp?id=35700261
“Non-portable systems are expected to hold the largest share of the fetal monitoring market, by portability in 2021.”
Based on portability, the market is segmented into non-portable systems and portable systems. In 2020, non-portable systems accounted for the largest share of this market, owing to wide applications in hospitals and diagnostic centers to assess fetal and maternal health.
“Noninvasive is expected to hold the largest share of the fetal monitoring market, by method in 2021.”
Based on the method, the market is segmented into invasive and noninvasive. In 2020, the noninvasive accounted for the largest share of this market. Noninvasive methods are widely accepted for their safer method of diagnosing and monitoring the fetus, especially in high-risk pregnancies. The rising approvals of various noninvasive monitoring devices and the incoming advanced technologies drive the growth of this segment.
“Antepartum is expected to hold the largest share of the fetal monitoring market, by application in 2021.”
Based on application, the fetal heart monitoring market is segmented into antepartum and intrapartum. The antepartum segment accounts for the largest share of the fetal monitoring market. Antepartum fetal monitoring is carried out throughout the gestation period to diagnose any congenital fetal abnormalities. The extensive monitoring regimes, recent product launches, surging prevalence of fetal anomalies, and maternal mortalities drive the growth of the antepartum segment.
“Hospitals are expected to hold the largest share of the fetal monitoring market, by end user in 2021.”
The market is segmented into hospitals, obstetrics & gynecology clinics, and home care settings based on end users. In 2020, hospitals accounted for the largest share of this market. This segments growth can be attributed to several factors, such as highly advanced facilities in hospitals and the increasing number of pregnancy procedures. Technological advancements, coupled with government funding, are also encouraging the installation of new tools and devices in hospitals, thus supporting market growth.
Download a PDF Brochure @ https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=35700261
“North America commanded the largest share of the fetal monitoring market in 2021.”
On the basis of region, the fetal monitoring market is segmented into North America, Europe, Asia Pacific, and the Rest of the World. In 2019, North America commanded the largest share of the fetal monitoring market. The large share of this market segment can be attributed to the presence of an established reimbufrsement structure coupled with availability of experts in the North American countries.
Leading Companies
The major players operating in this market are Cardinal Health, Inc. (US), Koninklijke Philips N.V. (Netherlands), GE Healthcare (US), Siemens Healthineers (Germany), FUJIFILM SonoSite, Inc. (US), Natus Medical Incorporated (US), Huntleigh Healthcare Limited (UK), The Cooper Companies Inc. (US), CONTEC Medical Systems Co., Ltd. (China), EDAN Diagnostics, Inc. (China), Neoventa Medical AB (Sweden), Bionet Co., Ltd. (South Korea), Progetti Srl (Italy), TRISMED Co., Ltd. (Republic of Korea), ShenZhen Luckcome Technology Inc. (China), MedGyn Products, Inc (US), Dixion (Germany), Promed Group Co., Ltd. (Hong Kong), Advanced Instrumentations (US), BRAEL-Medical Equipment (Poland), GPC Medical (India), BISTOS (Korea), Mediana (Korea), Life Plus (India), ChoiceMMed (China), Trivitron Healthcare (India), Dott Medical Co. Ltd. (China), Monarch Meditech (India), and Shenzhen Comen Medical Instruments Co., Ltd. (China).