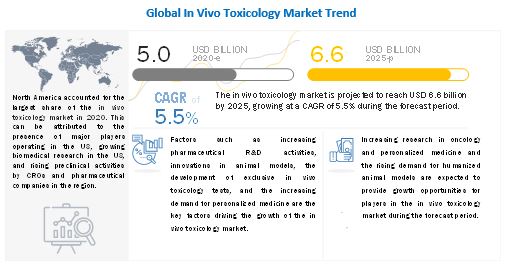
Growth Factors Driving the Market: The In Vivo Toxicology Market growth is driven largely by the increasing pharmaceutical R&D activities, innovations in animal models, the development of exclusive in vivo toxicology tests, and the increasing demand for personalized medicine
Rising demand for humanized animal models:
Humanized animal models are important tools for conducting preclinical research to gain insights into human biology. These models are developed through the engraftment of human cells or tissues, leading to the expression of human proteins in animals.
Humanized mice are increasingly being used as models for biomedical research applications, such as cancer, infectious diseases, HIV/AIDS, regenerative medicine, and hepatitis. In March 2019, the National Institute of Allergy and Infectious Diseases (NIAID), an agency of the US Department of Health and Human Services, announced funding for projects to conduct detailed characterization, direct comparisons, and further development of humanized immune system (HIS) mouse models. The need to identify the actual effects of drugs on humans, as well as the growing focus on studying human-specific infections, therapies, and immune responses, is promoting the development and use of humanized animal models
The global in vivo toxicology market size is projected to reach USD 6.6 billion by 2025 from USD 5.0 billion in 2020, at a CAGR of 5.5% during the forecast period.
Download PDF Brochure:
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=105308811

Industry Segmentation in Detailed:
Based on toxicity endpoint, the global in vivo toxicology market is segmented into immunotoxicity, systemic toxicity, carcinogenicity, genotoxicity, developmental & reproductive toxicity (DART), and other toxicity endpoints (includes organ toxicity, endocrine disruptor toxicity, juvenile toxicity, phototoxicity, ocular toxicity, and skin irritation). In 2019, the immunotoxicity segment accounted for the largest share of the global in vivo toxicology market, followed by the systemic toxicity segment.The rising demand for the development of biologics and biosimilars is driving the growth of the immunotoxicity segment
Based on test type, the global in vivo toxicology testing market is segmented into acute, sub-acute, sub-chronic, and chronic test type. The chronic test type segment is expected to dominate the market during the forecast period. In 2019, the chronic test type segment held the largest share of the market, followed by sub-chronic test type.Increasing research on drugs used for longer-duration therapy such as anti-cancer, anti-convulsive, anti-arthritis, and anti-hypertensives drives the growth of the chronic test type market.
Based on the testing facility, the global in vivo toxicology market is segmented into outsourced testing facilities and in-house testing facilities. The outsourced testing facilities segment is expected to dominate the market, by testing facility, over the forecast period. In 2019, the outsourced testing facilities segment held the largest share of the global in vivo toxicology market. The large share of this segment is attributed to the increasing R&D investments and cost-saving strategies of pharmaceutical, biopharmaceutical, and medical devices companies, resulting in increased outsourcing of services to CROs
Speak to Analyst: https://www.marketsandmarkets.com/speaktoanalystNew.asp?id=105308811
Leading Key Players Analysis:
The major players operating in In Vivo Toxicology Testing Market are by Charles River Laboratories (US), The Jackson Laboratory (US), Envigo (US), Taconic Biosciences, Inc. (US), and JANVIER LABS (France), Thermo Fisher Scientific (US), Danaher Corporation (US), Waters Corporation (US), Agilent Technologies (US), Shimadzu Corporation (Japan), Bruker Corporation (US), PerkinElmer (US). Other prominent players include Merck KGaA (Germany), GE Healthcare (US), and Bio-Rad Laboratories (US), genOway (France), Cyagen Biosciences (US), GVK BIO (India), PolyGene (Switzerland), Crown Biosciences (US), TransCure bioServices (France), Ozgene Pty Ltd. (Australia), Harbour BioMed (US) among others.
Geographical Trends in Detailed:
The global in vivo toxicology market is segmented into North America, Europe, the Asia Pacific, Latin Ametica and Middle East & Africa. In 2019, North America accounted for the largest share of the in vivo toxicology market. The large share of the North America region can be attributed to the presence of major players operating in the in vivo toxicology market in the US, growing biomedical research in the US, and rising preclinical activities by CROs and pharmaceutical companies in the region.
Request Research Sample Pages:
https://www.marketsandmarkets.com/requestsampleNew.asp?id=105308811

