Research Methodologies Followed for This Study:
Primary Research:
In the primary research process, various sources from both the supply and demand sides were interviewed to obtain qualitative and quantitative information for this report. The primary sources from the supply side include industry experts such as CEOs, vice presidents, marketing and sales directors, technology and innovation directors, and related key executives from various key companies and organizations operating in the pharmaceutical excipients market. The primary sources from the demand side include industry experts such as experts from drugs manufacturing companies, nutraceutical companies, and contract manufacturing organizations.
Secondary Research:
This research study involved the use of comprehensive secondary sources; directories and databases such as D&B, Bloomberg Business, and Factiva; and white papers, annual reports, and company house documents. Secondary research was used to identify and collect information for this extensive, technical, market-oriented, and commercial study of the global pharmaceutical excipients market. It was also used to obtain important information about the top players, market classification, and segmentation according to industry trends to the bottom-most level, geographic markets, technology perspectives, and key developments related to the market. A database of the key industry leaders was also prepared using secondary research.
COVID-19 Impact on Global Pharmaceutical Excipients Market
Covid-19 had a substantial impact on the pharmaceutical industry. Lockdowns that were imposed during the virus outbreak disrupted the supply chain for various pharmaceutical companies. These pharmaceutical drug manufacturing companies experienced shortage of raw materials such as excipients and APIs that further created a demand supply gap of drugs in the market. Additionally, regulatory authorities had to construct new laws to ensure maximum patient safety after drug consumption.
The end of the lockdowns saw the pharma industry gaining momentum, especially given the demand for drugs such as hydroxychloroquine and Remdesivir, which showed positive results against COVID-19. The increasing demand for these drugs drove revenue growth for some companies. The above mentioned factors suggest that the Covid-19 impact on the pharmaceutical excipients market is negligible as it is the starting material in manufacturing of pharmaceutical drugs.
Download PDF Brochure@
https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=956
Restraint: Expensive & time-intensive drug development process
According to the California Biomedical Research Association (CBRA), it takes an average of 12 years for a drug to travel from the research lab to the patient, including 1–6 years for toxicology studies alone. Also, only 5 in 5,000 drugs that begin preclinical testing ever make it to human testing. Of these, only one is approved for human usage. Moreover, late-stage failures in the drug development process increase R&D costs and impact the profit margins of pharmaceutical companies. Moreover, strict regulations on safety and efficacy of the drugs and raw materials such as excipeints create a need to enhance current quality assurance and manufactruing practices, which adds to the overall manufacturing cost. Therefore, even though drug development process has experienced considerable advances in past few years, extensive capital investments is expected to hamper market growth during the forecast period.
Opportunity: Emerging countries represent a great opportunity when it comes to pharmaceutical industry
Growth of pharmaceuticals industry has been restricted since a past few years due to government-induced pressure on reducing healthcare costs and the diminishing drug pipeline. This has encouraged the pharmaceutical manufacturing companies to expand their manufacturing bases in high-growth emerging markets. Manufacturers can leverage financial benefits such as low-cost of manufacturing in terms of attractive tax rates and lenient regulatory guidelines.
Challenge: Safety and quality concerns
With the current changes in government policies all over the world, upcoming trade policies could prove to be a challenge for the pharmaceutical industry, and in turn, the excipients industry. Trade between the developing countries who are considered to be pharmerging countries such as China, India, and Brazil and developed countries like the US and European countries, who are top markets in the industry, could be severely affected. BREXIT is another pressing issue in this market.
The possibility of fallout originating from Britain’s exit from the EU has led companies to consider shifting—or shift—their manufacturing from the UK to Germany, France, and other European countries. Although the effects of BREXIT on the pharmaceutical industry are still unclear, companies like Novartis, Astra Zeneca, and Roche have already shifted manufacturing sites in order to reduce any further risks.
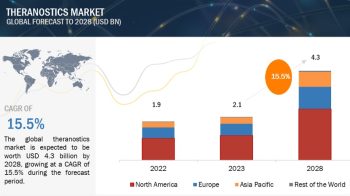
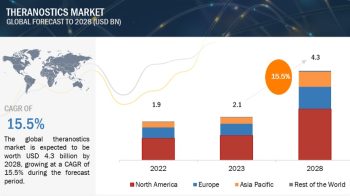
Europe region is expected to account for the largest share of the pharmaceutical excipeints market in 2020
In 2020, Europe accounted for the largest share of the pharmaceutical excipients market, followed by North America, the Asia Pacific, Latin America, and the Middle East & Africa. Several European countries are concentrating their attention on the generics market due to the expiration of blockbuster drug patents in the coming years. Due to this, the European region is expected to witness substantial growth in its generics market, which will in turn drive the requirement for pharmaceutical excipients. Rising government initiatives for lowering drug prices are expected to drive the market for generic drugs in these countries. Aforementioned factors, combined with growing investments for the development of biologics and advanced dosage forms, will increase the demand for novel excipients and thereby aid the growth of the pharmaceutical excipients market in Europe.
Request Sample Pages@
https://www.marketsandmarkets.com/requestsampleNew.asp?id=956